|
1/7/2023 0 Comments Metal ion bonding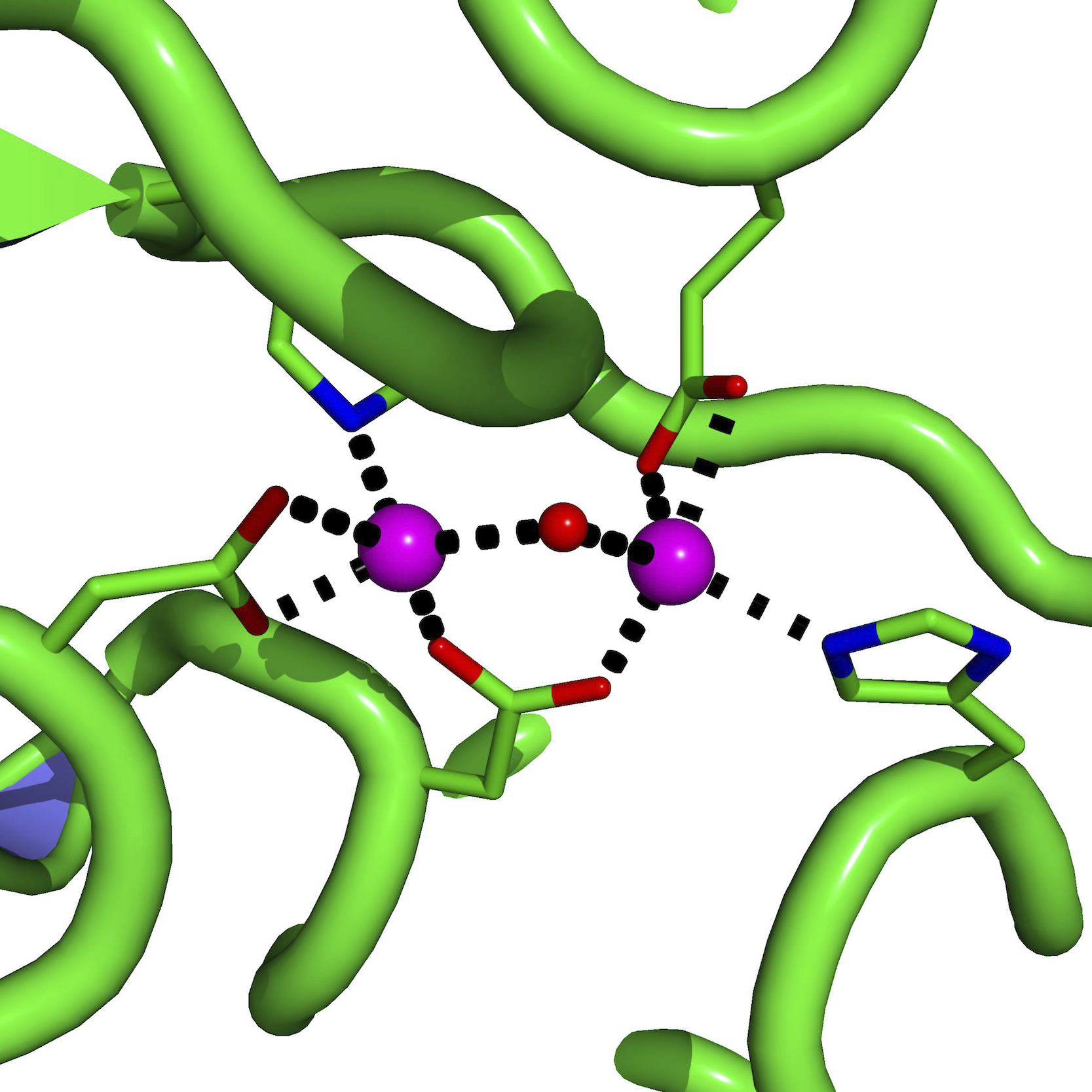
The irreducible representations that these span are a 1g, t 1u and e g. In molecular symmetry terms, the six lone-pair orbitals from the ligands (one from each ligand) form six symmetry adapted linear combinations (SALCs) of orbitals, also sometimes called ligand group orbitals (LGOs). Ligand-Field scheme summarizing σ-bonding in the octahedral complex 3+. In ligand field theory, the various d orbitals are affected differently when surrounded by a field of neighboring ligands and are raised or lowered in energy based on the strength of their interaction with the ligands. That is, the unoccupied d orbitals of transition metals participate in bonding, which influences the colors they absorb in solution. In their paper, they proposed that the chief cause of color differences in transition metal complexes in solution is the incomplete d orbital subshells. Griffith and Orgel used the electrostatic principles established in crystal field theory to describe transition metal ions in solution and used molecular orbital theory to explain the differences in metal-ligand interactions, thereby explaining such observations as crystal field stabilization and visible spectra of transition metal complexes. 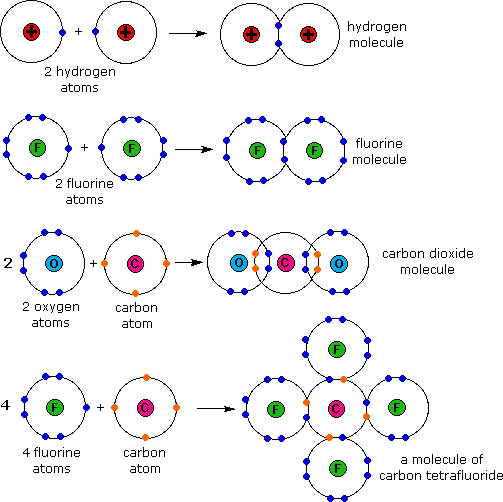 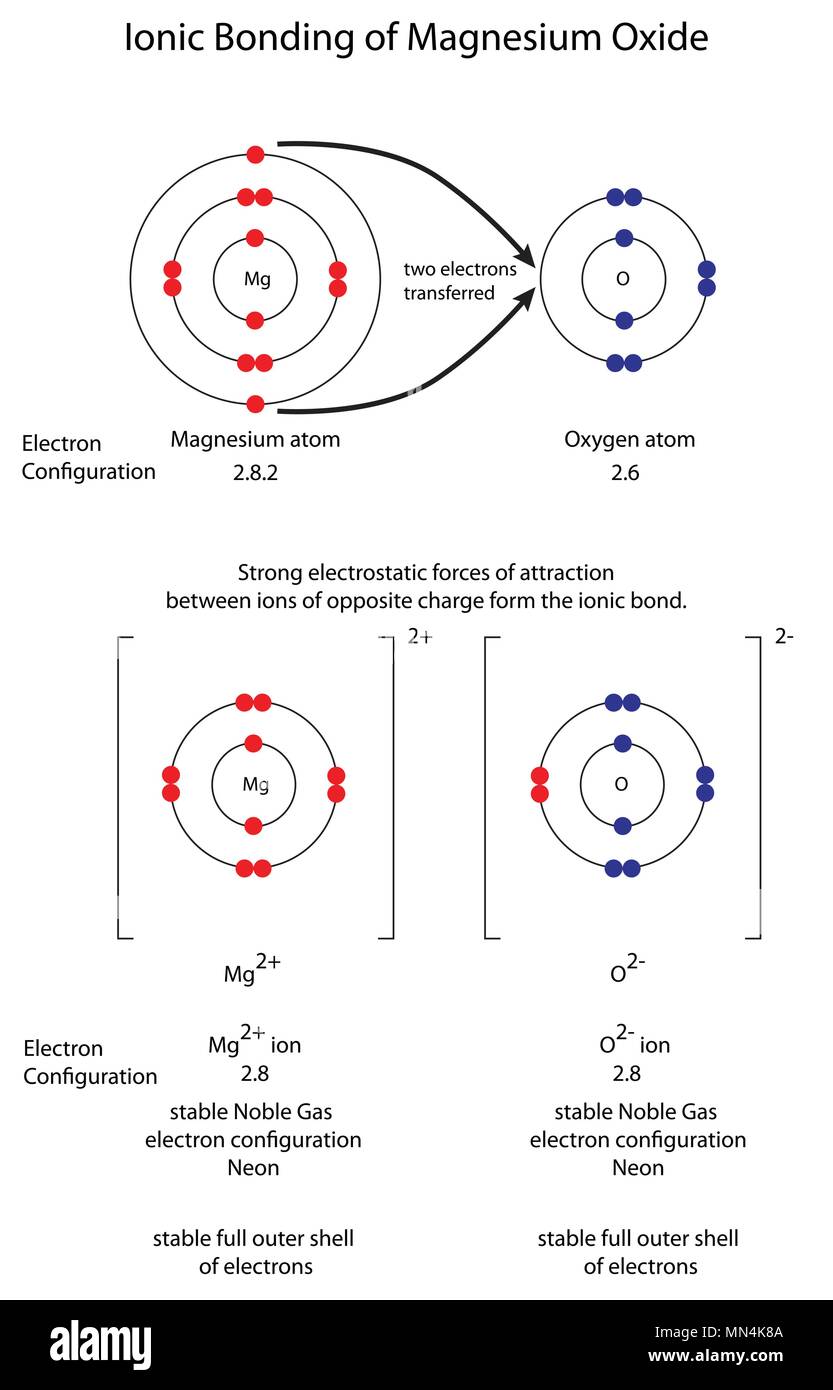
John Stanley Griffith and Leslie Orgel championed ligand field theory as a more accurate description of such complexes, although the theory originated in the 1930s with the work on magnetism of John Hasbrouck Van Vleck. Ligand field theory resulted from combining the principles laid out in molecular orbital theory and crystal field theory, which describes the loss of degeneracy of metal d orbitals in transition metal complexes. 3 High and low spin and the spectrochemical series.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
